Rutherford experiment conclusion6/11/2023  
What observations and conclusions did Rutherford recorded in the alpha particle scattering experiment? The size of the nucleus if very small as compared to the size of the atom.All the positive charge must be concentrated in a very small space within the atom called nucleus.Most of the space inside the atom is empty.What were the three major observations Rutherford made in the gold foil experiment? What are the 3 observations of Rutherford? Surprisingly very few α particles completely rebounded. Some α particles were deflected by small angles and a few by large angles. What are the observations and conclusions of Rutherford’s gold foil experiment?įrom the Gold foil experiment, Rutherford and his team observed that: Most of the fast moving α-particles passed straight through the gold foil. Some were deflected through small angles and some were deflected through large angles.He observed that most of the alpha particles passed straight through the gold foil.The observations made by Rutherford in his scattering experiment are as follows: There is a positive tiny part in the atom in its centre, which deflects or repels the α-particles. Therefore, most of the α-particles went through the gold foil without deflecting from their path. 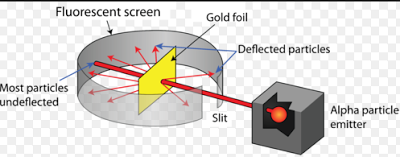
What are the conclusions of Rutherford’s?
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
